Email : [email protected]
URL : http://www.otcjournal.com
To
OTC Journal Members:
 |
Trading Alert-
QT 5 Inc. (OTC BB: QTFV) |
|
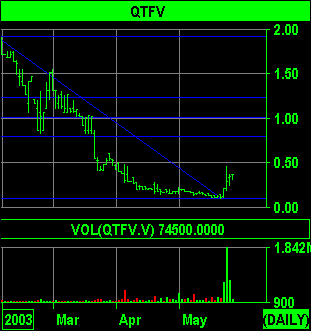
Last Thursday shares of microcap
stock QT5 inexplicably exploded up the charts on 10 times normal
volume. Over the past four months QT5 has drifted down from nearly
$2 to a mere $.10, averaging about 100,000 shares per day. Two trading
days later the stock peaked at 1.8 million shares and made a high of $.45,
a 450% move off the bottom.
Today, just as the market closed,
QT5
made
an announcement which clears up the mystery surrounding the resurgence
of interest in the stock. QT5 announced their highly anticipated
product has been made available at a store near you, and investors are
buying up shares as quickly as product is rumored to be disappearing off
the shelves.
QT5 revealed its highly anticipated
smoking substitute, NICOWater, had been rolled out for sale on
a nationwide basis. NICOWater is a homeopathic formula developed
for adult smokers who suffer from the symptoms of tobacco cravings and
find themselves in situations and/or environments where smoking is prohibited
or discouraged.
QT5 has introduced a non carcinogenic
beverage which satisfies the craving for a cigarette. The size of the market
is staggering.
 |
NICOWater-
The Product and Market |
 |

Pictured here is a display you will
probably be able to find in your neighborhood grocery store or pharmacy
chain in the near future. NICOWater is not to be confused with
a smoking cessation product. It does not replace a patch or a gum designed
to help people quit smoking.
NICOWater is a smoking substitute.
It is a beverage you can enjoy in any environment where smoking is prohibited
or discouraged. Smokers can drink this beverage in bars, restaurants, airplanes,
and offices. It satisfies their craving for a cigarette without offending
others.
It is estimated there are still 47
million smokers in the United States. In 1985 there were only 200 municipalities
with clean air ordinances. Today there are 1609, and the number is growing
at the municipal, state, and nationwide levels. Massachusetts is the most
recent state to ban smoking in public buildings.
Anti smoking forces have set a target
date of 2010 to ban smoking in every public building in the United States.
Therefore, the 47 million Americans who still smoke will need to satisfy
their craving without spending too much time on the sidewalk or suffering
on a five hour plane ride. Bars and restaurants would prefer to have their
customers stay on their property. Employers want employees to stay at their
desks. Airlines want another source of revenue in the form of a beverage
they can sell to a captive audience for the duration of a flight to a happy
traveler.
Americans spend approximately $200
billion annually on tobacco products. 1% of the market would equate to
$2 billion in annual sales, which seems overly optimistic. 1/10th of 1%
out of the gates seems a more realistic expectation, which still equates
to the possibility of $20 million in sales for the near term.
 |
High
Profile Publicity |
 |

Investors pounced on shares of QT5
because the product has already generated a great deal of high profile
publicity, and there are high expectations for sales. The media has set
investor expectations high for major product acceptance in the early stage
of introduction.
In fact, NICOWater has already
been the subject of major coverage in the main stream media including:
-
USA Today
-
CNBC
-
CNN
-
The Wall Street Journal
-
Time Magazine
-
The New York Times
-
Newsweek
-
Business Week
-
The LA Times
-
Forbes
This is only a partial list of the publicity
the product generated before it was introduced. As it appears on shelves
in stores, expect to read or hear about NICOWater in all sorts
of nationwide and regional media. A major nationwide marketing campaign
is planned, which includes regional advertising like the sign shown on
the taxi cab.
QT5 has established relationships
with 6 regional bottlers covering every major metropolitan area in the
United States, each of whom has the capability to bottle approximately
1 million bottles per month.
Initially, the company is focusing
its marketing energy on Chain Drug outlets. Over the next several weeks
I expect the company to issue several press releases disclosing the identity
of initial customers on the size of initial orders. I hope to publish information
on projected revenues and profit margins. Perhaps you will be able to visit
a store near you and see the product on display.
 |
Conclusion |
 |
Last week QT5 rocketed from
$.10 to a high trade of $.45 in just three trading days as the rumor spread
that this long awaited product was finally being shipped to stores.
I believe this stock has just begun
to capture the imagination of investors. As the product appears in stores
around the nation, and sales figures begin coming in, this stock will probably
gain momentum. After all, the size of the market is staggering and there
is virtually no competition. Gums and patches are smoking cessation products
used by those wishing to quit tobacco. NICOWater is a smoking substitute
people will use in places where smoking is prohibited or discouraged.
There are only 32 million shares
issued and outstanding, giving the company a market value of about $10
million at the $.30 level. However, only 8 million of those 32 million
shares are publicly traded, which could lead to dramatic price swings if
demand far outpaces supply.
The stock has pulled back to about
$.30 after last week's prolific surge to $.45, which in nearly a perfect
50% retracement of the move. Taking into account that sales are just beginning,
and the potential high profile publicity this product could generate, here
is my recommendation:
-
Buy QTFV up to about $.40.
-
Target price- The stock is entitled
to rebound into the $1 range as the midpoint of its four month drop. Therefore,
I am setting a more moderate target of $.75 (the low end of the
range) over the next 30 days. By then we will be able to gauge the momentum
of product sales and re-evaluate.
-
Stop Loss- $.20- With the upside
here definitely worth the risk of 33% to 50% of the capital you invest
in this one.
I hope to see several news releases
on the product roll out in the near future, and intend to publish follow
up editions if and when they come out.
Here is the complete text of today's
press release for your review:
| Press Release Source: QT 5, Inc.
QT 5, Inc. Announces National
Launch and Rollout of NICOWater(TM)
Wednesday May 28, 4:01 pm ET
Multi-Million Dollar Development Program Culminates
in Breakthrough Homeopathic Product
WESTLAKE VILLAGE, Calif., May 28
/PRNewswire-FirstCall/ -- QT 5, Inc. (OTC Bulletin Board: QTFV - News),
manufacturer and marketers of NICOWater(TM), the breakthrough Homeopathic
nicotinum (nicotine) product designed to relieve symptoms of tobacco cravings,
announced today the national launch and roll-out NICOWater(TM). NICOWater(TM),
the result of a multi-million dollar investment program over a year in
development, is now being rolled out nationally.
"This is a very exciting threshold
moment in QT 5's history as this revolutionary Homeopathic product -- over
which the management, employees and our partners have labored intensively
for a year -- now comes to market," stated Steven Reder, QT 5's President.
"Our breakthrough product -- specifically targeting the extraordinarily
attractive $200 billon market for tobacco and tobacco products -- has been
engineered to satisfy smokers' cravings when they can't smoke or shouldn't
smoke, an increasingly common dilemma for smokers in today's restrictive
public and private environments." Mr. Reder went on to say, "QT 5 is populated
by a dynamic management team which has defined a compelling opportunity
to capitalize on a market populated by over 25% of all Americans. Our burgeoning
company, which sustained significant losses in the development and manufacturing
of this state-of-the-art Homeopathic product, will now begin to reap the
rewards of its labor by addressing the needs of these millions of American
smokers."
About QT 5, Inc.
QT 5, Inc. is a Delaware corporation
formed in April 1999 as a manufacturer, distributor and marketer of Bio-Med
testing and Homeopathic products. QT 5, Inc. is continuing its clinical
research and development of future products for lifestyle enhancements.
QT 5, Inc. is headquartered in Westlake Village, California. For more information
please visit: www.qt5inc.com. NICOWater(TM) is sold under the FDA's Compliance
Policy Guide Conditions Under Which Homeopathic Drugs May Be Marketed.
For additional information on QT 5, Inc. contact Bernadette Cusack, Vice
President -- Investor Relations, Trilogy Capital Partners, [email protected].
Cautionary Statement
This release contains forward-looking
statements that involve risks and uncertainties, such as statements about
our plans, objectives, expectations, assumptions or future events. These
statements involve estimates, assumptions, known and unknown risks, uncertainties
and performances, or achievements expressed or implied by the forward-looking
statement. Actual future results and trends may differ materially from
those made in or suggested by any forward-looking statements due to a variety
of factors, including, for example, our ability to compete with other products
in our space; the risk of unfavorable federal regulation; and the fact
that our status as a development state company makes our future unclear.
Consequently you should not place undue reliance on these forward-looking
statements. We discuss many of these and other risks and uncertainties
in greater detail under the section entitled, "Risk Factors That May Affect
Future Results" in Item 1 of the Company's Current Report on Form 8-K as
filed with SEC on January 24, 2003 (the "Merger Form 8-K").
--------------------------------------------------------------------------------
Source: QT 5, Inc. |
Charts Provided Courtesy
Of TradePortal.com |